Vasa previa is an obstetrical complication associated with
fetal death and neonatal morbidity and is defined as fetal velamentous vessels
overlying or in close proximity to the cervix. Because these velamentous
vessels lack the protection of Wharton jelly or the placenta and are fixed
within the membranes, they are at risk of laceration during labor or membrane
rupture with resultant fetal exsanguination. Another cause of fetal compromise
with vasa previa is postulated to be compression of the velamentous vessels, leading
to fetal asphyxia.
When undiagnosed prenatally, vasa previa is associated with
perinatal mortality as high as 56% and significant morbidity in survivors. The
risk of perinatal death and hypoxic-ischemic morbidity is 25-fold and 50-fold
greater, respectively, than in cases in which vasa previa is diagnosed during
the antepartum period. With prenatal diagnosis with ultrasound and an
appropriate surveillance and management strategy, the perinatal mortality rate
is < 1%. Intact survival with prenatal diagnosis is also significantly
higher with prenatal diagnosis than in those without a prenatal diagnosis.
While prenatal diagnosis is associated with a reduction in
perinatal mortality rate, infants are still at risk of complications related to
prematurity. An important management tenet to reduce the risk of morbidity and
mortality is the delivery of the fetus before the onset of labor, usually in
the late preterm period. In this study, the factor most associated with
unplanned delivery before scheduled cesarean was multifetal gestation, and the
most common reasons for unplanned delivery were preterm labor/contractions,
followed by vaginal bleeding and nonreassuring fetal testing.
A metaanalysis of 113 cohort studies and case series including
1298 pregnancies with prenatally diagnosed vasa previa determined that
perinatal mortality is rare when vasa previa is diagnosed prenatally, affecting
0.5% of cases. In this metaanalysis, only 0.51% of the mortalities were related
to a ruptured vasa previa. Congenital anomalies or prematurity accounted for
the majority of deaths.
Diagnosis
From a historical perspective, vasa previa was suspected
when a patient presented with vaginal bleeding, and an Apt test indicated that
the blood was fetal in origin before the wide availability of ultrasound. The
Apt test is acolorometric alkaline denaturation test. Because fetal hemoglobin
is more resistant to denaturation than adult hemoglobin, a fetal specimen will
turn pink when mixed with sodium hydroxide whereas maternal hemoglobin will
turn yellow-brown. The Apt test is no longer available in the United States and
has been replaced by the vastly superior technique for vasa previa diagnosis
using ultrasound.
In this modern era, vasa previa is an ultrasound diagnosis
confirmed by transvaginal ultrasound demonstrating velamentous fetal vessels
unprotected by Wharton jelly or placenta within 2 to 5 cm from the internal os.
The differential diagnosis includes funic presentation, placenta previa,
membrane separation, subchorionic hematoma, and maternal vascular sinuses.
On gray-scale imaging, a vasa previa will appear as an
echogenic line or a thin-walled echogenic oval with a central echolucency. In
many cases, findings associated with vasa previa can be subtle when using
gray-scale imaging alone. However, the application of color Doppler to the area
will demonstrate flow behind the echogenic line or within the echogenic oval.
Pulsed Doppler will demonstrate either venous or arterial flow. If the arterial
flow is present, the rate will be consistent with the fetal heart rate. If
venous flow is demonstrated, tracing the vessel to its origin and destination
will assist in identifying it as a fetal vessel.
To differentiate vasa previa from alternative diagnoses, the
following techniques are helpful:
Evaluate the vessel(s) in orthogonal planes and trace the
vessels using ultrasound to their origins—both in the placenta and in the
fetus.
Use color Doppler to demonstrate flow within the area.
Apply pulsed Doppler to determine the heart rate (if
arterial vessel).
Three dimensional ultrasound rendering the coronal view of
the lower uterine segment while color Doppler is applied is useful in identifying
lateral velamentous vessels and demonstrating the relationship of these vessels
to the internal cervical os. Asking the
patient to cough, manually elevating the fetal presenting part, and/or
adjusting the head of the examination table may assist in differentiating funic
presentation from vasa previa.
Role of Screening for Vasa Previa
Controversy exists over who and how to screen for vasa
previa. Because prenatal diagnosis of vasa previa is lifesaving, many experts
advise universal screening for vasa previa. A focus group involving 68
international experts using a Delphi process concluded that all pregnancies
should be screened for vasa previa using ultrasound at the time of the
second-trimester anatomy scan.
For practices that have implemented universal cervical
length screening with transvaginal ultrasound at the time of the anatomical
ultrasound to assess the risk of spontaneous preterm birth, screening for vasa
previa means close inspection of the area immediately surrounding the internal
os for signs of vasa previa using gray scale ultrasound and color Doppler.
Alternatively, a 2-step screening process has been proposed by which: Transabdominal ultrasound is used to inspect
the placental edge, placental cord insertion, and lower uterine segment.
If any of the
following is noted—succenturiate or multilobate placenta, velamentous cord
insertion, multifetal gestation, low placenta (defined as previa or low lying),
or in-vitro fertilization pregnancy, then an abdominal scan of the cervix with
color Doppler is performed. Transvaginal ultrasound is recommended if the
transabdominal ultrasound cannot visualize the cervix sufficiently or there is
concern for vasa previa. When vasa previa or a low placenta is detected with
ultrasound screening, a follow-up ultrasound should be performed at 28 to 32
weeks to ensure that the placenta has moved away from the cervix and that there
is no vasa previa remaining after placental migration.
Management
Antepartum Surveillance
Asymptomatic patients diagnosed with vasa previa on
ultrasound and no risk factors for spontaneous preterm birth can reasonably be
managed as outpatients with monthly ultrasound for fetal growth due to the
association of vasa previa with fetal growth restriction. In practice, authors
recommend pelvic rest and avoidance of sexual intercourse. Serial cervical
surveillance with transvaginal ultrasound assessment of cervical length may be
considered to evaluate the patient’s risk of spontaneous preterm birth. If vasa
previa is again seen on transvaginal ultrasound at 28 to 32 weeks, a discussion
should occur with the patient regarding inpatient observation.
Many experts recommend routine inpatient observation as
early as 32 weeks with close monitoring for contractions and fetal assessment
with frequent nonstress testing. Inpatient observation has also been proposed
to allow close proximity to an operating room should the patient experience
vaginal bleeding, preterm prelabor membrane rupture, or demonstrate signs of
fetal decompensation on electronic fetal monitoring. A disadvantage of routine
hospitalization may be an increased risk of venous thromboembolism and
iatrogenic preterm birth.
For patients carrying a singleton gestation without signs of
vaginal bleeding, contractions, or cervical shortening, continued outpatient
management may be considered with weekly biophysical profiles and non-stress
testing. Shared decision-making should be used to determine inpatient versus
outpatient management and the timing of inpatient admission (if inpatient
management is agreed upon). For those patients at higher risk of spontaneous or
indicated preterm delivery, practice is to manage them with inpatient
observation. Patients carrying a singleton gestation with vaginal bleeding,
persistent preterm contractions, or short cervix in the third trimester are
admitted at the time of symptom or finding onset. Uncomplicated multifetal
gestations are typically admitted for inpatient management at 32 weeks; they
are admitted earlier in the third trimester if they present with vaginal
bleeding, persistent preterm contractions, or short cervix.
As the vast majority of patients with prenatally diagnosed
vasa previa will be delivered preterm, consideration should be given to
antenatal corticosteroid administration to reduce the risk of neonatal
morbidity related to prematurity. There is no consensus on the timing of
corticosteroid administration but it should be based on patient history, signs
and symptoms, and clinical judgment. Steroids are often administered at the
time of hospitalization (before 34 wk gestation) as indications for urgent
delivery may preclude steroid administration.
Delivery
Delivery for patients with uncomplicated vasa previa is
recommended through cesarean delivery between 35 and 37 weeks. Within this
range, 36 weeks appears to achieve the best perinatal outcomes when balancing
the risks of prematurity against the risk of a sudden unpredictable fetal
event. For patients with multifetal gestations, short cervix, and/or signs or
symptoms of preterm labor, the timing of delivery should be individualized through
shared decision-making.
Authors advise the following approach to performing cesarean
delivery in the setting of vasa previa:
One unit of O-negative packed red blood cells should be
ordered and held in the operating room for the neonate in the event of unanticipated
fetal vessel rupture and bleeding during fetal extraction efforts.
Once neuraxial anesthesia has been introduced and the
patient has been positioned for the cesarean, ultrasound mapping of the fetal
vessels in the lower uterine segment is performed (and it may be performed
preoperatively or intraoperatively).
In the absence of a complicated web of fetal vessels
overlying the anterior lower uterine segment, a low transverse uterine incision
is planned through a space avoiding any velamentous vessels.
When the low transverse uterine incision is made, all
attempts are made to maintain intact membranes until the uterine incision is
expanded and a safe window for delivery of the fetus is identified.
The lower uterine segment is elevated using allis clamps to
avoid inadvertent membrane rupture during uterine incision.
Once the uterus is entered, the incision is sharply expanded
laterally using bandage scissors with the membranes protected by the surgeons’
fingers between the membranes and the lower uterine segment.
The membranes are not ruptured until an area free of fetal
vessels is safely identified, and the membranes are entered sharply.
In practice, authors do not perform delayed cord clamping
for pregnancies affected by vasa previa as it is difficult to determine whether
the velamentous vessels remain intact after delivery of the infant.
Role of Fetoscopic Laser Ablation
Fetoscopic laser ablation of fetal vessels has been
investigated as a treatment of vasa previa with the goal of resolving the vasa
previa thus allowing the patient to achieve a term vaginal birth. It is a
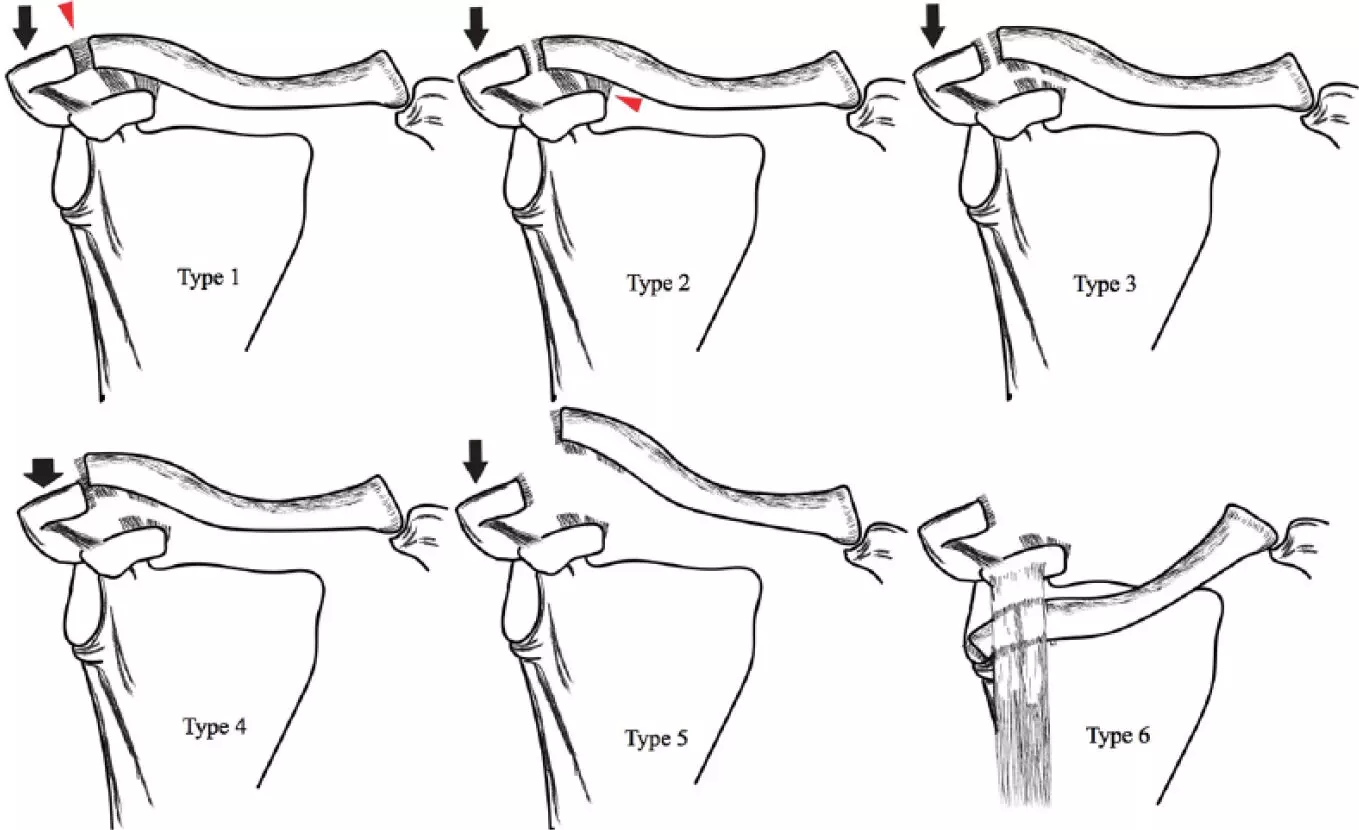
procedure that has been proposed only for the treatment of type 2 and 3 vasa
previa as type I vasa previas involves major vascular branches arising directly
from the umbilical cord. There have been several case series describing the
procedure and outcomes. The most recent one involved fetoscopic laser ablation
of 20 vasa previa cases at 31 to 33 weeks and demonstrated a mean delivery
gestational age of 37.2 weeks and a 70% vaginal delivery rate. There were no
perioperative complications in this series. However, operative fetoscopy, in
general, is associated with preterm prelabor rupture of membranes, preterm
delivery, and placental abruption, among other complications. At this time,
fetoscopic laser ablation of vasa previa therapy is considered experimental,
and larger-scale research studies are necessary before implementing it into
routine practice.
Vasa previa is an uncommon finding associated with a
substantial risk of perinatal death and morbidity when undiagnosed prenatal.
Implementation of routine ultrasound screening for vasa previa in the second
trimester is associated with >95% intact perinatal survival. Optimal
outcomes are achieved with a surveillance approach involving shared
decision-making with the patient and delivery through cesarean between 35 and
37 weeks for otherwise uncomplicated vasa previa patients.
Source: Ross and Roman; Clin Obstet Gynecol Volume 68, Number 1, March 2025