Generation X, millennials in US have higher risk of developing 17 cancers compared to older generations
Powered by WPeMatico
Powered by WPeMatico
Powered by WPeMatico
Powered by WPeMatico
Powered by WPeMatico

Ahmedabad: Zydus Lifesciences Limited has announced that the Mexican regulatory authority COFEPRIS (Federal
Commission for the Protection Against Sanitary Risk), has granted marketing approval for
Mamitra, a Trastuzumab biosimilar.
The drug will be marketed in different strengths of 150
mg and 440 mg and used in the treatment of patients with HER2 overexpressing metastatic
breast cancer (MBC), HER2 overexpressing early breast cancer (EBC) and advanced gastric
cancer. Breast cancer has become the most diagnosed cancer in Mexico, overtaking prostate
and colorectal cancers.
Speaking on the development, Dr. Sharvil Patel, Managing Director, Zydus said, “The approval
of Mamitra in Mexico allows us to expand the reach of our biosimilar portfolio to newer
markets and enable access to affordable life-saving therapies, particularly in oncology. Our
commitment to science, health and innovation helps us bridge unmet healthcare needs of
patients globally.”
Zydus had developed and launched the Trastuzumab biosimilar developed in-house by the
research team at the Zydus Research Centre (ZRC) in 2016 in India under the brand name
Vivitra. Since then, an estimated 1,00,000 patients have been treated with the therapy.
Read also: Zydus Life Sciences Gets CDSCO Panel Nod to Study anti-cancer drug Relugolix
Zydus Lifesciences Ltd. is an innovative, global lifesciences company that discovers, develops, manufactures, and markets a broad range of healthcare therapies. Zydus has been actively discovering and developing New Chemical Entities (NCEs) novel biologicals, several biosimilars and vaccines as a part of its innovation pipeline. Over the last decade, Zydus has introduced several products in the market for treating unmet healthcare needs with vaccines, therapeutics, biologicals and biosimilars.
Read also: Zydus Lifesciences wins USFDA okay for hypertension drug Valsartan
Powered by WPeMatico
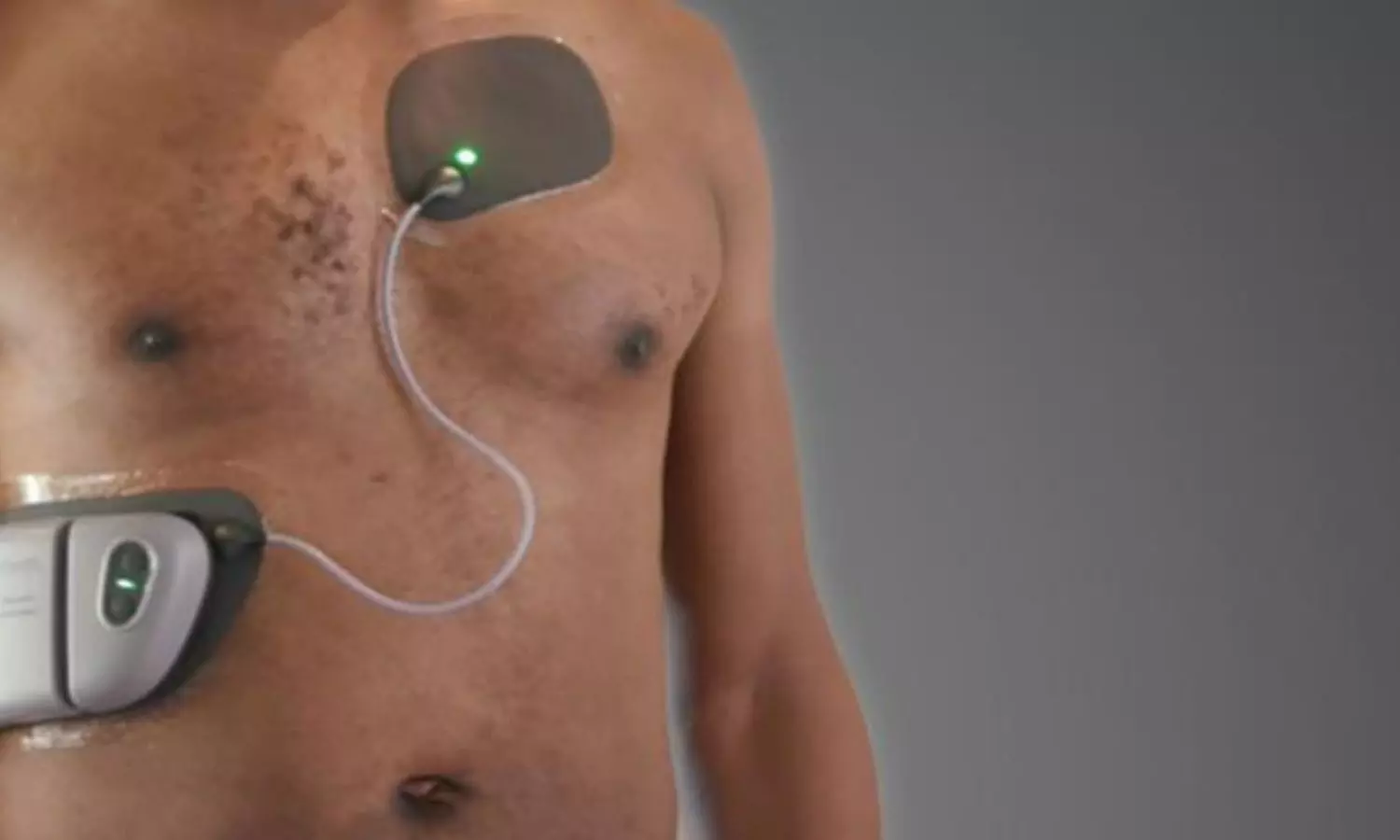
USA: A Jewel patch-wearable cardioverter-defibrillator effectively administered life-saving shocks to prevent sudden cardiac arrest (SCA), and users demonstrated high adherence to wearing the water-resistant device, a recent study has shown.
The Jewel is an innovative, water-resistant patch-wearable cardioverter-defibrillator (P-WCD) featuring a machine-learning detection algorithm aimed at enhancing both user compliance and protection against SCA.
“The innovative patch-wearable cardioverter-defibrillator is both safe and effective, with excellent patient adherence. There were no fatalities linked to non-compliance, and it achieved a high rate of successful shock conversions,” the researchers wrote in the Journal of the American College of Cardiology.
For many patients, the risk of sudden cardiac arrest is heightened temporarily. Wearable cardioverter-defibrillators (WCDs) can monitor and address SCA during these critical periods. However, traditional WCDs often come with discomfort, frequent maintenance needs, and limitations such as being unsuitable for use while showering, leading to issues with compliance and preventable SCA-related deaths.
Against the above background, John Hummel, The Ohio State University Wexner Medical Center, Columbus, Ohio, USA, and colleagues aimed to demonstrate the safety and clinical effectiveness of a novel P-WCD.
For this purpose, the researchers enrolled patients at SCA risk due to ventricular tachycardia/ventricular fibrillation who were not candidates for or refused an implantable defibrillator in the Jewel IDE Study, a prospective, single-arm study conducted at 30 U.S. sites.
The main safety endpoint was set as less than 15% of patients experiencing clinically significant cutaneous adverse effects from the device, while the primary effectiveness endpoint was fewer than two inappropriate shocks per 100 patient months. Secondary endpoints included achieving at least one successful conversion of ventricular tachycardia or ventricular fibrillation and maintaining a wear time compliance of over 14.1 hours per day.
The following were the key findings of the study:
“This study of a new, water-resistant patch-wearable cardioverter-defibrillator (P-WCD) successfully achieved both primary and secondary effectiveness and safety endpoints. There were no patient deaths or missed episodes that required external rescue, and the high level of patient compliance resulted in a substantial number of successful, life-saving conversions,” the researchers concluded.
Reference:
Hummel, J., Houmsse, M., Tomassoni, G., Nair, D., Romero, J., Hargrove, J., Mathews, K., Thakkar, A. B., Ullery, S., Eapen, Z. J., Kumar, U. N., Mehran, R., & Butler, J. (2024). A Patch Wearable Cardioverter-Defibrillator for Patients at Risk of Sudden Cardiac Arrest. Journal of the American College of Cardiology, 84(6), 525-536. https://doi.org/10.1016/j.jacc.2024.04.063
Powered by WPeMatico
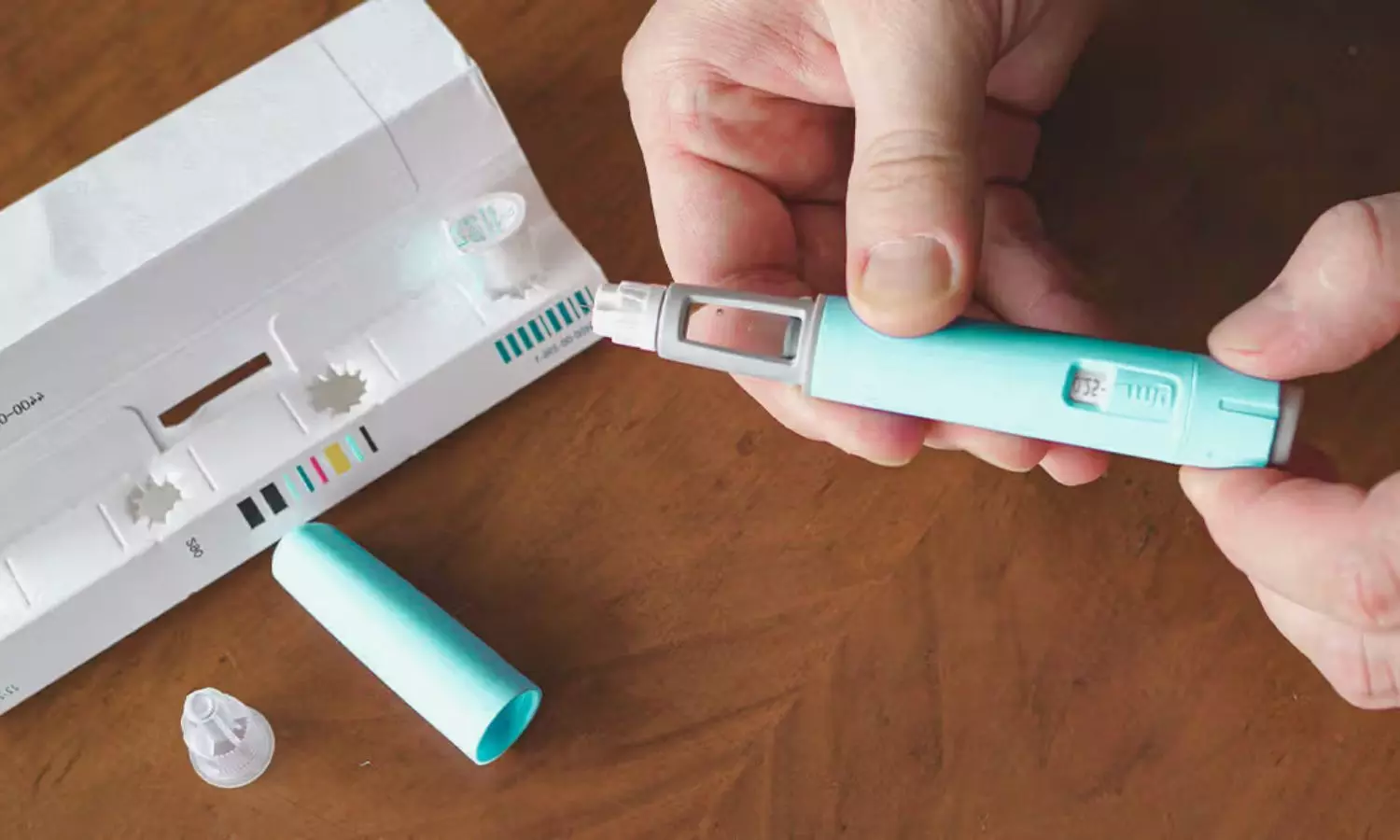
USA: Patients are experiencing overdoses of compounded semaglutide due to errors in both self-administration and clinician calculations, which may differ from those for FDA-approved products.
On July 26, the FDA issued a warning after receiving reports of dosing mistakes with compounded semaglutide injectable products dispensed from multidose vials. These errors have led to adverse effects such as gastrointestinal issues, fainting, dehydration, headaches, gallstones, and acute pancreatitis, with some patients requiring hospitalization.
FDA-approved semaglutide products are standardized in milligram dosages and come only in prefilled pens, ensuring consistency and reducing the risk of dosing errors. In contrast, compounded semaglutide products may vary significantly. They can come in multidose vials or prefilled syringes, and their concentrations can differ depending on the compounder. Some compounders may offer multiple concentrations, complicating accurate dosing.
Semaglutide is a medication classified as a glucagon-like peptide-1 (GLP-1) receptor agonist. Currently, there are three FDA-approved semaglutide products:
Instructions for compounded drugs may specify doses in “units” rather than milligrams, and these units can vary with concentration. This discrepancy can lead to patients receiving syringes with volumes much larger than prescribed.
The FDA has documented instances where patients inadvertently took doses from multidose vials that were 5 to 20 times higher than prescribed. Errors often occur when clinicians miscalculate doses during conversions from milligrams to units or milliliters. For example, one patient, unable to clarify dosing instructions from a telemedicine provider, sought online advice and ended up taking five times the intended dose.
In another case, a clinician prescribed 20 units instead of the correct dose of 2 units, leading to three patients receiving ten times the intended dose and experiencing symptoms like nausea and vomiting. Additionally, a clinician, who also used semaglutide, mistakenly administered a dose ten times higher than intended.
The FDA has previously cautioned about the risks associated with compounded drugs, especially during shortages like that of semaglutide. While compounded drugs can be beneficial in some cases, they generally pose a higher risk than FDA-approved drugs because they do not undergo the same rigorous premarket review for safety, effectiveness, or quality.
“FDA recognizes the substantial consumer interest in using compounded semaglutide products for weight loss. However, compounded drugs pose a higher risk to patients than FDA-approved drugs because compounded drugs do not undergo FDA premarket review for safety, effectiveness, or quality,” the FDA stated.
“Therefore, compounded drugs should only be used to meet a patient’s needs if they cannot be met by an available FDA-approved drug,” the agency concluded.
Powered by WPeMatico

A new study published in the recent edition of the Nature Mental Health journal found that juvenile type 1 diabetes is associated with elevated risk of anxiety, behavioral syndromes, mood, personality problems and other mental health issues.
Type 1 diabetes (T1D) results from the death of pancreatic β-cells and a permanent lack of insulin. There is strong evidence from several large-scale population-based studies that T1D with a juvenile beginning and ensuing adult mental problems are significantly comorbid. When compared to their classmates, children with T1D report higher levels of discomfort, more problems in behavior and a worse quality of life. Also, parents of these children with T1D report significant levels of family disturbance and suffering. Therefore, to overcome the shortcomings of previous research and to evaluate putative causal links between mental problems and childhood-onset T1D, Tomáš Formánek and team undertook this study.
A two-step analytical method was employed in this work to reduce bias and/or confounding. First, the relationship between T1D and later psychiatric illnesses was examined using national registration data from the Czech Republic. Also, the data from extensive genome-wide association studies (GWAS) involving European individuals were subjected to separate bidirectional two-sample Mendelian randomization (MR) analysis in order to further examine the connections.
According to the findings of this study, kids with T1D were more likely to experience behavioral syndromes, mood, anxiety, personality problems and drug abuse. On the other hand, the children with T1D were less likely to experience psychotic episodes. A relationship with schizophrenia was also detected in a Mendelian randomization study. However, this association did not hold after multiple testing corrections. Together, observational and Mendelian randomization data point to a predisposing relationship between childhood T1D diagnosis and significant, far-reaching psychological illness that is unlikely to be explained by shared underlying biological processes.
Overall, the outcomes of this research highlighted the extensive and far-reaching psychological effects of a juvenile T1D diagnosis by combining in-depth observational and genetic epidemiology investigations. In the decades that follow a childhood T1D diagnosis, there is an increased chance of acquiring most mental diseases, with the exception of psychoses, according to this observational research.
Source:
Formánek, T., Chen, D., Šumník, Z., Mladá, K., Hughes, J., Burgess, S., Wareham, N. J., Murray, G. K., Jones, P. B., & Perry, B. I. (2024). Childhood-onset type 1 diabetes and subsequent adult psychiatric disorders: a nationwide cohort and genome-wide Mendelian randomization study. In Nature Mental Health. Springer Science and Business Media LLC. https://doi.org/10.1038/s44220-024-00280-8
Powered by WPeMatico
An experimental drug originally developed to treat cancer may help clear HIV from infected cells in the brain, according to a new Tulane University study.
For the first time, researchers at Tulane National Primate Research Center found that a cancer drug significantly reduced levels of SIV, the nonhuman primate equivalent of HIV, in the brain by targeting and depleting certain immune cells that harbor the virus.
Published in the journal Brain, this discovery marks a significant step toward eliminating HIV from hard-to-reach reservoirs where the virus evades otherwise effective treatment.
“This research is an important step in tackling brain-related issues caused by HIV, which still affect people even when they are on effective HIV medication,” said lead study author Woong-Ki Kim, PhD, associate director for research at Tulane National Primate Research Center. “By specifically targeting the infected cells in the brain, we may be able to clear the virus from these hidden areas, which has been a major challenge in HIV treatment.”
Antiretroviral therapy (ART) is an essential component of successful HIV treatment, maintaining the virus at undetectable levels in the blood and transforming HIV from a terminal illness into a manageable condition. However, ART does not completely eradicate HIV, necessitating lifelong treatment. The virus persists in “viral reservoirs” in the brain, liver, and lymph nodes, where it remains out of reach of ART.
The brain has been a particularly challenging area for treatment due to the blood-brain barrier — a protective membrane that shields it from harmful substances but also blocks treatments, allowing the virus to persist. In addition, cells in the brain known as macrophages are extremely long-lived, making them difficult to eradicate once they become infected.
Infection of macrophages is thought to contribute to neurocognitive dysfunction, experienced by nearly half of those living with HIV. Eradicating the virus from the brain is critical for comprehensive HIV treatment and could significantly improve the quality of life for those with HIV-related neurocognitive problems.
Researchers focused on macrophages, a type of white blood cell that harbors HIV in the brain. By using a small molecule inhibitor to block a receptor that increases in HIV-infected macrophages, the team successfully reduced the viral load in the brain. This approach essentially cleared the virus from brain tissue, providing a potential new treatment avenue for HIV.
The small molecule inhibitor used, BLZ945, has previously been studied for therapeutic use in amyotrophic lateral sclerosis (ALS) and brain cancer, but never before in the context of clearing HIV from the brain.
The study, which took place at the Tulane National Primate Research Center, utilized three groups to model human HIV infection and treatment: an untreated control group, and two groups treated with either a low or high dose of the small molecule inhibitor for 30 days. The high-dose treatment lead to a notable reduction in cells expressing HIV receptor sites, as well as a 95-99% decrease in viral DNA loads in the brain .
In addition to reducing viral loads, the treatment did not significantly impact microglia, the brain’s resident immune cells, which are essential for maintaining a healthy neuroimmune environment. It also did not show signs of liver toxicity at the doses tested.
The next step for the research team is to test this therapy in conjunction with ART to assess its efficacy in a combined treatment approach. This could pave the way for more comprehensive strategies to eradicate HIV from the body entirely.
Reference:
Diana G Bohannon, Laurent D Zablocki-Thomas, Evan S Leung, Jinbum K Dupont, Julian B Hattler, Jolanta Kowalewska, Miaoyun Zhao, Jiangtao Luo, Marco Salemi, Angela M Amedee, Qingsheng Li, Marcelo J Kuroda, Woong-Ki Kim. CSF1R inhibition depletes brain macrophages and reduces brain virus burden in SIV-infected macaques. Brain, 2024; DOI: 10.1093/brain/awae153.
Powered by WPeMatico
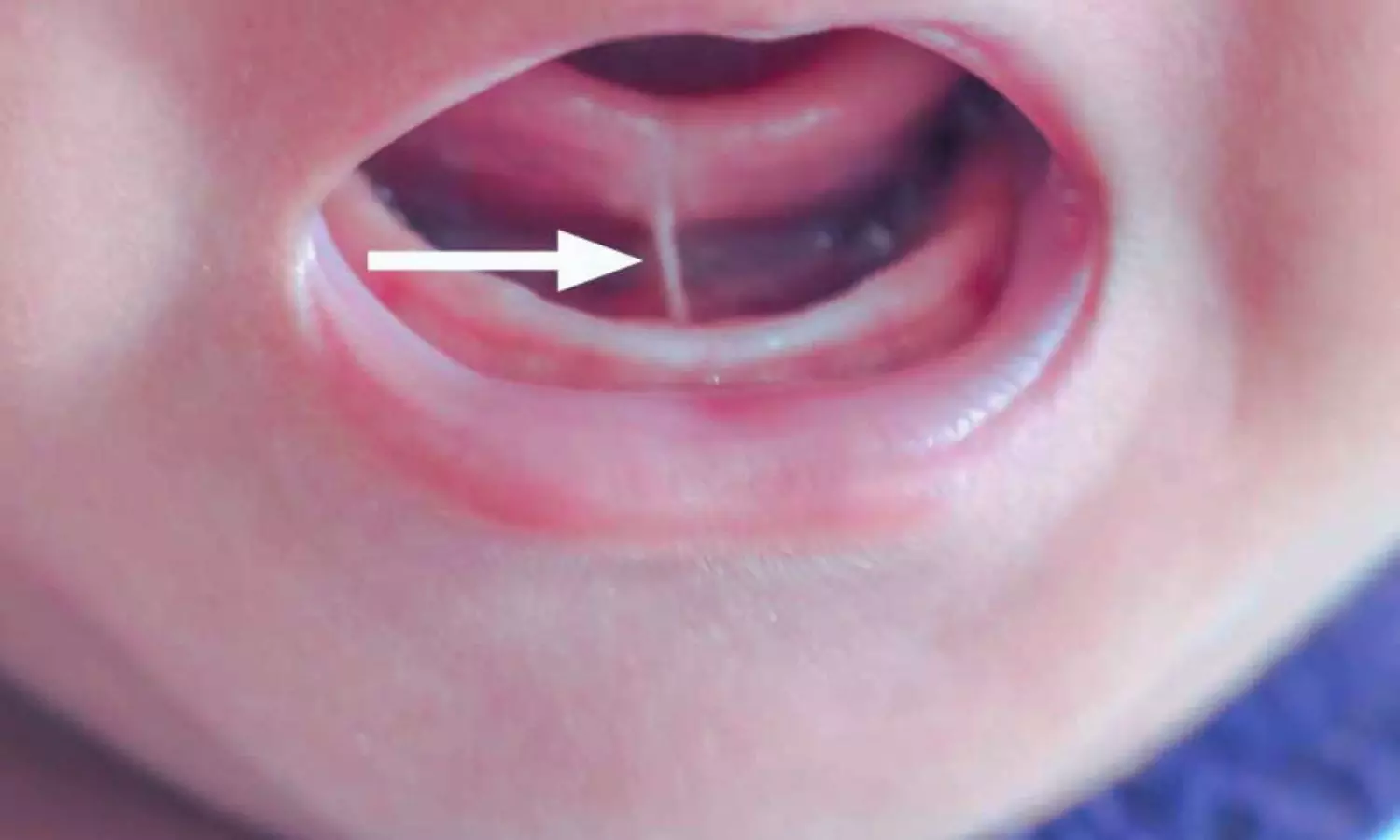
USA: The American Academy of Pediatrics (AAP) has released a clinical report and recommendations on identifying and managing ankyloglossia and its effect on breastfeeding in infants.
According to the recommendation published in Pediatrics, pediatricians and other specialists have an important role in identifying ankyloglossia, a condition present at birth and commonly referred to as tongue-tie, and its effect on breastfeeding in infants.
While the AAP recommends exclusive breastfeeding for about six months with possible continuation for two years or longer, ankyloglossia can impede breastfeeding, Jennifer Thomas, Department of Pediatrics, Advocate Aurora Health, Milwaukee, Wisconsin, and colleagues noted.
Ankyloglossia, commonly known as tongue-tie, “limits the tongue’s movement due to a short or tight band of tissue called the lingual frenulum, which connects the underside of the tongue to the floor of the mouth,” the authors explained. This restriction “can hinder breastfeeding by preventing the infant from extending and lifting their tongue effectively to latch onto the nipple and suckle properly.”
They wrote, “Currently, there are no standardized diagnostic criteria for the condition, and treatment approaches remain unsettled. Despite this, both diagnoses and the frequency of performing frenotomy—a procedure that involves cutting the lingual frenulum—have recently seen an increase.”
A series of studies conducted in the U.S., Canada, and England have revealed that the rate of tongue-tie diagnoses in newborns varies between 1.7% and 10.7%. In the U.S., diagnoses surged nearly tenfold from 1997 to 2012, with a further doubling from 2012 to 2016. This trend was mirrored by an increase in frenotomy procedures.
Although several tools for assessing ankyloglossia severity have been published, none have been validated, according to Thomas and his colleagues. Recent reviews of small randomized trials on frenotomy also failed to demonstrate whether the procedure leads to long-term breastfeeding success or alleviates maternal pain.
The report’s recommendations include that infants with ankyloglossia who are feeding normally should not undergo intervention. Additionally, frenotomy is not supported for preventing future issues like speech problems or obstructive sleep apnea. Experts also do not agree on posterior tongue-tie as a valid reason for surgery, and labial and buccal frenae, as well as sucking blisters, are considered normal and do not require surgical intervention.
For cases of inadequate breastfeeding, a multidisciplinary approach involving lactation specialists, feeding therapists, surgeons, and pediatricians is crucial for optimal outcomes. Newborns with symptomatic ankyloglossia should receive breastfeeding support in the hospital, follow-up care post-discharge, and monitoring of weight gain. Surgical options, including frenotomy versus laser treatment, should be considered only after evaluating and addressing other breastfeeding issues. While the evidence is limited, frenotomy might reduce maternal nipple pain, contributing to successful breastfeeding.
Thomas and his team stress that frenotomy should be performed by a trained professional who provides a comprehensive discussion of alternatives, risks, benefits, pain control options, and post-surgical care. They also emphasize the importance of preventing surgical complications, managing hemorrhage risk, and avoiding evidence-free practices such as stretching exercises.
The team concluded that further research is needed to establish standardized methods for diagnosing and classifying ankyloglossia, assess long-term outcomes, and understand the natural history of the condition and its risks to feeding.
Reference:
Jennifer Thomas, Maya Bunik, Alison Holmes, Martha Ann Keels, Brenda Poindexter, Anna Meyer, Alison Gilliland, SECTION ON BREASTFEEDING, SECTION ON ORAL HEALTH, COUNCIL ON QUALITY IMPROVEMENT AND PATIENT SAFETY, COMMITTEE ON FETUS & NEWBORN, SECTION ON OTOLARYNGOLOGY-HEAD AND NECK SURGERY; Identification and Management of Ankyloglossia and Its Effect on Breastfeeding in Infants: Clinical Report. Pediatrics 2024; e2024067605. 10.1542/peds.2024-067605
Powered by WPeMatico
