COVID-19 no longer means five days in isolation, CDC says
Powered by WPeMatico
Powered by WPeMatico
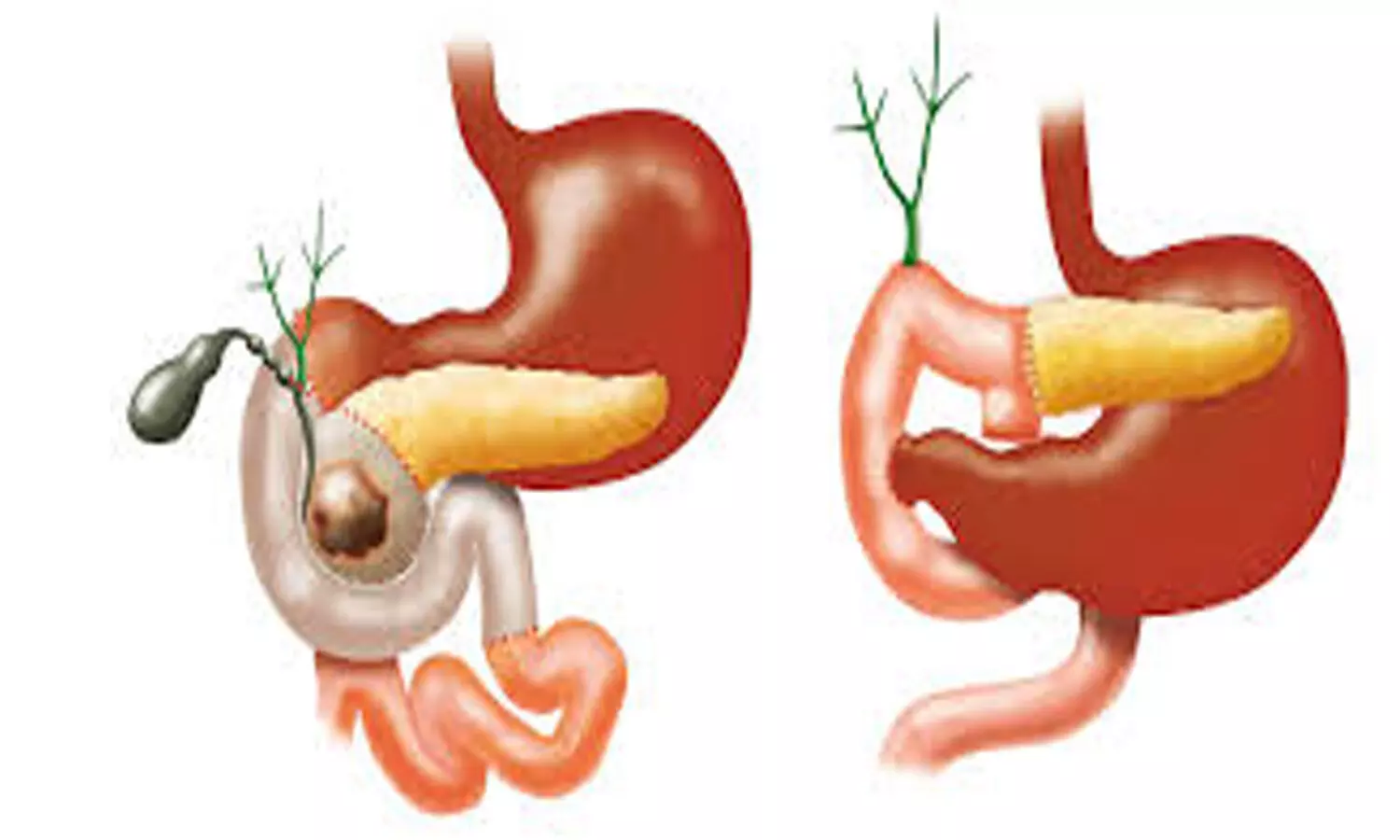
A recent open-label randomized trial conducted in China assessed the feasibility and safety of robot-assisted pancreatoduodenectomy compared to open surgery for pancreatic head or periampullary tumors. The study aimed to evaluate short-term outcomes, including postoperative hospital length of stay, operating times, blood loss, and complications. The study was published in the journal Lancet Gastroenterology and Hematology by Prof Qu Liu and colleagues.
Pancreatoduodenectomy is a complex surgical procedure commonly used to treat pancreatic and periampullary tumors. Robot-assisted surgery has gained attention as a potential alternative to open surgery, offering potential advantages such as shorter hospital stays and reduced blood loss. However, conclusive evidence regarding its efficacy and safety compared to traditional open surgery has been limited.
The multicenter trial involved 164 patients randomly assigned to either robot-assisted or open pancreatoduodenectomy. Highly experienced surgeons performed the procedures, with five surgeons from high-volume hospitals participating. The study evaluated short-term outcomes, including postoperative hospital length of stay, operating times, blood loss, complications, readmission rates, and mortality.
Key Findings:
Robot-assisted pancreatoduodenectomy significantly reduced postoperative hospital length of stay compared to open surgery (median 11 vs. 13.5 days).
Robot-assisted surgery also demonstrated shorter operating times (245 vs. 298 minutes) and reduced blood loss (75 vs. 150 mL).
Complication rates, readmission rates, and mortality were similar between the two groups.
Pathology outcomes, including tumor characteristics, were comparable between the robotic and open surgery groups.
The trial concluded that robot-assisted pancreatoduodenectomy is feasible and safe, with modest improvements in short-term outcomes compared to open surgery. While the study focused on short-term benefits, the long-term oncological outcomes require further investigation.
Despite the promising results, the generalizability of the findings may be limited, and the clinical benefit of robot-assisted surgery should be carefully weighed against the associated costs. Future research should prioritize assessing long-term oncological outcomes and maintaining comprehensive registries for outcome tracking in robot-assisted pancreatoduodenectomy.
Reference:
Liu Q, et al “Effect of robotic versus open pancreaticoduodenectomy on postoperative length of hospital stay and complications for pancreatic head or periampullary tumours: a multicentre, open-label randomised controlled trial” Lancet Gastroenterol Hepatol 2024; DOI: 10.1016/S2468-1253(24)00005-0. https://www.thelancet.com/journals/langas/article/PIIS2468-1253(24)00005-0/abstract
Powered by WPeMatico

The US Food and Drug Administration (FDA) has approved Exblifep® (cefepime/enmetazobactam) for treatment of complicated urinary tract infections (cUTIs), including pyelonephritis among patients 18 years of age and older.
Allecra has also received a five-year marketing exclusivity extension from the FDA as part of the Generating Antibiotic Incentives Now Act (GAIN Act). The GAIN Act, enacted by the U.S. Congress, incentivizes the creation of new anti-infective therapeutics by providing benefits to manufacturers of Qualified Infectious Disease Products (QIDPs).
“Receiving FDA approval is a tremendous achievement for Allecra and a testament to the hard work and dedication of a small, yet highly focused team of individuals. I extend my sincere congratulations to my colleagues Omar Lahlou and Patrick Velicitat for their leadership and oversight throughout this whole process,” said Iain Buchanan, Supervisory Board Member of Allecra Therapeutics. “As we continue our discussions with strategic partners for product launch in the U.S., we value the FDA’s positive decision on EXBLIFEP®’s ability to address a critical unmet medical need for patients.”
The FDA’s approval of EXBLIFEP® was supported by a totality of clinical data that demonstrated EXBLIFEP® effectiveness against antimicrobial resistance in gram-negative bacteria, especially resistance mediated by both ESBL (Extended Spectrum Beta Lactamases) and AmpC. This included results from Allecra’s Phase 3 ALLIUM trial, which met criteria for non-inferiority and superiority compared to piperacillin/tazobactam in the primary composite outcome of clinical cure and microbiological eradication in patients with cUTIs.
Allecra is the sole holder of a significant patent estate covering EXBLIFEP® in major territories with the GAIN Act extending Allecra’s market exclusivity until 2032. Enmetazobactam was first discovered by Orchid Pharma and all rights outside India were assigned to Allecra Therapeutics in 2013. The company has since taken the sole responsibility for the international clinical and regulatory development of EXBLIFEP®. Allecra was founded through a strategic partnership formed by Nicholas Benedict, Stuart Shapiro and Edward Currie in conjunction with Orchid Chemicals and Pharmaceuticals Ltd. and Allecra lead investors, Andera Partners, Forbion and EMBL Ventures. The company has concluded exclusive license agreements for EXBLIFEP® with Shanghai Haini Pharmaceutical in Greater China and ADVANZ PHARMA in Europe.
EXBLIFEP® (cefepime/enmetazobactam) has been investigated in patients with complicated urinary tract infections (cUTIs) compared to piperacillin/tazobactam, a current standard of care, in a randomized, controlled, double-blind, global Phase 3 trial. EXBLIFEP® has already been submitted for Marketing Authorization Approval in Europe by Allecra’s commercial partner, Advanz Pharma. The European Medicines Agency (EMA) has indicated that, in light of results obtained in an epithelial lining fluid penetration study, the company is eligible for approval of cefepime/enmetazobactam for use in hospital-acquired/ventilator-associated bacterial pneumonia. A positive opinion was received from the CHMP (The Committee for Medicinal Products for Human Use) in January 2024.
Powered by WPeMatico

Chronic inflammation has been increasingly identified as a potential risk factor for VTE. While research on this topic is ongoing, it is essential to note that limited data is available on the risk of VTE in patients with sarcoidosis. This condition affects the immune system and causes granuloma formation.
Powered by WPeMatico

The impact of abdominal fat on brain health and cognition is generally more pronounced in middle-aged men at high risk of Alzheimer’s disease as opposed to women, according to researchers at Rutgers Health.
In middle-aged individuals with a family history of Alzheimer’s disease, the amount of fat in their abdominal organs (pancreas, liver, and belly fat) is related to their brain volumes and cognitive function, according to the study published in the journal Obesity. The study was written by Sapir Golan Shekhtman, a Ph.D. student at the Joseph Sagol Neuroscience Center at the Sheba Medical Center in Israel and led by Michal Schnaider Beeri, director of the Herbert and Jacqueline Krieger Klein Alzheimer’s Research Center at Rutgers Brain Health Institute.
The research, conducted on 204 healthy middle-aged Alzheimer’s-dementia offspring, investigated fat depots in the pancreas, liver and abdomen measured with MRI.
“In middle-aged males at high Alzheimer’s disease risk-but not females-higher pancreatic fat was associated with lower cognition and brain volumes, suggesting a potential sex-specific link between distinct abdominal fat with brain health,” said Beeri, who is the Krieger Klein Endowed Chair in Neurodegeneration Research at BHI and a faculty member of the Rutgers Institute for Health, Health Care Policy and Aging Research.
Obesity is a risk factor for lower cognitive functioning and higher dementia risk, with different associations between sexes.
The research findings highlight the importance of investigating the interrelationships of fat depots, brain aging and cognition in the context of sex differences.
Additionally, the study challenges the conventional use of body mass index (BMI) as the primary measure for assessing obesity-related cognitive risks. The researchers said BMI poorly represents body fat distribution and does not necessarily account for sex differences.
“Our findings indicate stronger correlations compared to the relationships between BMI and cognition, suggesting that abdominal fat depots, rather than BMI, is a risk factor for lower cognitive functioning and higher dementia risk,” said Shekhtman.
These research findings open new avenues for targeted interventions and further exploration of sex-specific approaches in understanding and mitigating the impact of abdominal fat on brain health, Shekhtman noted.
Reference:
Golan Shekhtman S, Boccara E, Ravona-Springer R, Inbar Y, Zelicha H, Livny A, Bendlin BB, Lesman-Segev O, Yore I, Heymann A, Sano M, Mardor Y, Azuri J, Schnaider Beeri M. Abdominal fat depots are related to lower cognitive functioning and brain volumes in middle-aged males at high Alzheimer’s risk. Obesity (Silver Spring). 2024 Feb 27. doi: 10.1002/oby.24004.
Powered by WPeMatico

Researchers have found in a new study that Telehealth medication abortion is effective, safe and comparable to published rates of in-person medication abortion care. The large national study finds that video visits, texting and mailing pills are all effective, as the U.S. Supreme Court considers limiting access to telemedicine abortion.
Medication abortion can be delivered safely and effectively through telemedicine, according to new research from UC San Francisco that comes as the U.S. Supreme Court is about to hear a case that could severely restrict access to one of the two pills that are used to induce abortions.
Researchers analyzed data from more than 6,000 patients who obtained abortion pills from virtual clinics in 20 states and Washington D.C. between April of 2021 and January of 2022. They found there were no serious adverse events 99.8% of the time, and that abortions did not require follow-up care 98% of the time. This is similar to what has been found for patients who receive medication abortion at clinics or doctor’s offices.
The findings appear Feb. 15, 2024, in Nature Medicine.
Medication abortion, which now accounts for more than half of all abortions, involves taking two pills – mifepristone followed by misoprostol – and extensive research supports its safety and efficacy. Demand for the pills has increased since more than two dozen states banned or restricted abortion following the June 2022 decision by the Supreme Court to overturn Roe v. Wade, which guaranteed a constitutional right to abortion.
On March 26, the Supreme Court is scheduled to hear arguments over whether the U.S. Food and Drug Administration (FDA), which approved mifepristone in 2000, should roll back rules that in recent years have made the medication more widely available. In 2021, the agency removed the in-person dispensing requirement, allowing health care providers to have abortion pills sent through the mail after an online consultation. Telehealth abortion now accounts for nearly 10% of all U.S. abortions.
“This research confirms that the FDA followed science in allowing patients to get medication abortion through telehealth and the mail,” said the study’s first author, Ushma Upadhyay, PhD, MPH, a public health scientist with Advancing New Standards in Reproductive Health (ANSIRH) at UCSF. “Our findings make it abundantly clear that the case at the Supreme Court is simply an attempt to restrict access to abortion care, even in those states where it’s legal.”
The researchers also compared video visits to secure text messaging, and found they were about equally safe and effective. The authors concluded that telehealth protects patient privacy while making abortion more accessible. This has become increasingly important as abortion clinics have closed in states that restricted abortion and wait times have increased elsewhere.
“Since the COVID-19 pandemic, telemedicine has become the new frontier in health care, including for medication abortion,” said Upadhyay, who is a professor of Obstetrics, Gynecology & Reproductive Sciences at UCSF. “A ruling against this method and the FDA’s rigorous science review process would be a huge blow to the American public and make this essential health service harder to get.”
The study has already been cited in the amicus brief submitted to the Supreme Court supported by more than 300 leading reproductive health researchers.
Reference:
Upadhyay, U.D., Koenig, L.R., Meckstroth, K. et al. Effectiveness and safety of telehealth medication abortion in the USA. Nat Med (2024). https://doi.org/10.1038/s41591-024-02834-w
Powered by WPeMatico

Rheumatoid arthritis impacts approximately 2 million people in the United States and is associated with increased risk of cardiovascular disease. However, assessing cardiovascular risk is difficult in patients with rheumatoid arthritis because standard clinical assessments based on factors like age, cholesterol, and smoking status tend to underestimate cardiovascular risk in individuals with rheumatoid arthritis.
In a new study published in the Journal of the American Heart Association, a research team led by physicians at Mass General Brigham with expertise in rheumatology and cardiovascular disease identified six blood biomarkers that are associated with cardiovascular risk in patients with rheumatoid arthritis and whose measurements improved the researchers’ ability to predict a future increase in arterial inflammation. The biomarkers hold the potential to clinically assess an individual patient’s risk of cardiovascular disease, but more research is needed to determine whether they are associated with cardiovascular events such as heart attack or stroke.
“We think these biomarkers might improve our ability to predict risk and intervene early to help our patients,” said first author Daniel H. Solomon, MD, MPH, chief of the Section of Clinical Sciences in the Division of Rheumatology and Matthew H. Liang Distinguished Chair at Brigham and Women’s Hospital, a founding member of the Mass General Brigham healthcare system. “The idea is that if we measure biomarkers that are specific to rheumatoid arthritis, we might be able to better identify those at highest risk of cardiovascular events.”
To identify rheumatoid arthritis-specific biomarkers of cardiovascular risk, the researchers assembled a panel of 24 candidate biomarkers that had been previously shown to be associated with rheumatoid arthritis and systemic inflammation. Then, they measured the concentration of these biomarkers in 109 patients with rheumatoid arthritis who were taking part in a randomized clinical trial (the TARGET Trial) to compare the efficacy of two different treatments for rheumatoid arthritis at preventing cardiovascular disease. The researchers measured the biomarkers at the beginning of the study and six months later, imaging the patients’ arteries at each time to assess their arterial inflammation—an indicator of cardiovascular risk.
“Arterial inflammation can predict future cardiovascular disease risk,” said cardiologist and co-author Ahmed Tawakol, MD, the director of Nuclear Cardiology and co-director of the Cardiovascular Imaging Research Center at Massachusetts General Hospital, a founding member of the Mass General Brigham healthcare system. “If you take a snapshot of a person’s blood vessels, the more inflammation that is measured there, the greater the likelihood the person will have progression of their disease, and the greater likelihood that they will have a stroke or a myocardial infarction.”
Six of the 24 biomarkers were associated with increased cardiovascular risk and using them in predictive models improved the researchers’ ability to predict increases in arterial inflammation compared to standard clinical indices such as the Framingham Risk Score, which is based on factors such as age, sex, cholesterol, blood pressure, diabetes, and smoking.
“This is an important step towards using blood samples to measure changes in cardiovascular risk with the treatment of rheumatoid arthritis,” said Solomon.
The study showcases the strength of ongoing collaborations between Brigham and Women’s Hospital and Massachusetts General Hospital, said Solomon and Tawakol, who trained together as residents at the Brigham around 30 years ago. “Having two really great institutions collaborating in the same organization meant we could leverage the strengths of the respective institutions and teams,” said Solomon.
Now, the team is working to test these biomarkers in a larger and more long-term cohort of rheumatoid arthritis patients, the Brigham and Women’s Rheumatoid Arthritis Sequential Study (BRASS), which has been following over 1,000 patients with rheumatoid arthritis since 2003. This follow-up study will allow the researchers to not only test associations between the biomarkers and arterial inflammation, but also assess whether the biomarkers can predict future cardiovascular events such as heart attack or stroke.
Reference:
Daniel H. Solomon, Olga Demler, Pamela M. Rist, Leah Santacroce, Ahmed Tawakol, Jon T. Giles, Katherine P. Liao and Joan M. Bathon, Biomarkers of Cardiovascular Risk in Patients With Rheumatoid Arthritis: Results From the TARGET Trial, Journal of the American Heart Association, https://doi.org/10.1161/JAHA.123.032095.
Powered by WPeMatico

Plant-based diets are becoming popular in Germany and Western countries because of sustainable lifestyles, animal welfare, and health concerns. A flexitarian diet, which emphasizes plant-based foods while allowing for small amounts of meat and processed meat products, attracts followers who primarily cite health benefits as their motivation.
According to a BMC Nutrition study, a flexitarian diet is associated with lower cardiovascular risk than an omnivorous diet.
More data on CVD risk factors’ relationship with flexitarian diets must be collected. This study included 94 healthy participants aged 25-45 years, who were divided into three groups: long-term flexitarians (FXs), vegans (Vs), and omnivores (OMNs). Metabolic biomarkers, body composition, blood pressure, arterial stiffness ( measured by pulse wave velocity, PWV) and metabolic syndrome (MetS) severity were measured. Dietary intake, diet quality, and physical activity levels were also assessed.
Key findings from the study are:
· Compared to omnivores, Flexitarians and vegans had more beneficial insulin levels, triglycerides, total cholesterol, and LDL cholesterol.
· Flexitarians had the most favourable MetS-score results based on BMI and waistline and better PWV values compared to vegans and omnivores.
· FXs and Vs had higher intake rates of vegetables, fruit, nuts/seeds and plant-based milk alternatives.
Diet impacts CVD risk. Meat and processed meat intake correlate with an unfavourable risk profile.
Study limitations include cross-sectional design, small sample size, and potential recall bias.
Study strengths include well-controlled design, homogeneity, and additional CVD risk indicators such as MetS scores and PWV.
This study highlights that plant-based diets improve blood lipid profiles and have higher diet quality. The results of this study demonstrate the beneficial impact of a flexitarian diet on CVD risk parameters.
Reference:
Bruns, A., Greupner, T., Nebl, J. et al. Plant-based diets and cardiovascular risk factors: a comparison of flexitarians, vegans and omnivores in a cross-sectional study. BMC Nutr 10, 29 (2024). https://doi.org/10.1186/s40795-024-00839-9
Powered by WPeMatico
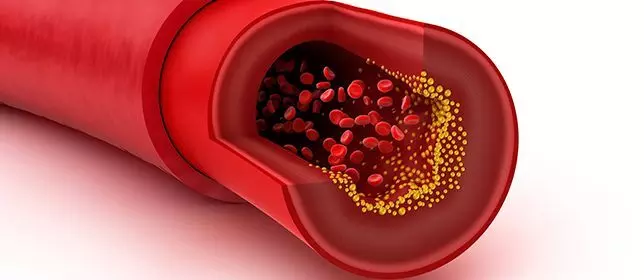
A recent study unveiled a crucial link between Lipoprotein(a) [Lp(a)] levels and the risk of atherosclerotic cardiovascular disease (ASCVD). The findings were published in the Journal of the American College of Cardiology and explored the intricate relationship between Lp(a) and major adverse cardiovascular events (MACE).
The study was conducted across two medical centers in Boston, Massachusetts, analyzed data from over 16,000 individuals that spanned nearly two decades to determine the association between Lp(a) levels and the incidence of MACE among patients with and without pre-existing ASCVD. This research utilizing Cox proportional hazards modeling and categorized participants into Lp(a) percentile groups and evaluated their risk of MACE, including nonfatal myocardial infarction (MI), nonfatal stroke, coronary revascularization or cardiovascular mortality.
The results from the analysis found that out of the 62% of patients with baseline ASCVD, the individuals in the 71st to 90th percentile Lp(a) group expressed a notable 21% increased hazard of MACE, close to those in the 91st to 100th percentile group. Also, among the remaining participants without established ASCVD, there was a progressive increase in MACE risk with rising Lp(a) levels, with the individuals in the highest percentile group facing a sharp 93% relative risk increase. These findings challenge conventional risk assessment paradigms suggests that the optimal Lp(a) threshold for gauging cardiovascular risk may differ based on the individuals in the primary or secondary prevention cohorts.
Reference:
Berman, A. N., Biery, D. W., Besser, S. A., Singh, A., Shiyovich, A., Weber, B. N., Huck, D. M., Divakaran, S., Hainer, J., Kaur, G., Blaha, M. J., Cannon, C. P., Plutzky, J., Januzzi, J. L., Booth, J. N., III, López, J. A. G., Kent, S. T., Nasir, K., Di Carli, M. F., … Blankstein, R. (2024). Lipoprotein(a) and Major Adverse Cardiovascular Events in Patients With or Without Baseline Atherosclerotic Cardiovascular Disease. In Journal of the American College of Cardiology (Vol. 83, Issue 9, pp. 873–886). Elsevier BV. https://doi.org/10.1016/j.jacc.2023.12.031
Powered by WPeMatico
Germany: A randomized clinical trial suggests that intraoperative wound irrigation with polyhexanide solution should not be recommended as standard clinical practice in open clean-contaminated surgical procedures in patients undergoing laparotomy.
In the study published in JAMA Surgery, the researchers did not find a significant difference in the rates of surgical site infection (SSI) between intraoperative wound irrigation with polyhexanide compared to saline or no irrigation in 689 patients undergoing laparotomy.
Postoperative SSI is the most common type of hospital-acquired infection across all income and development settings following gastrointestinal surgery. Surgical site infections increase costs, mortality and morbidity rates and prolong hospital stay. Intraoperative wound irrigation is used worldwide as a preventive measure, although evidence supporting this practice is lacking.
To fill this knowledge gap, Tara Catharina Mueller, Technical University of Munich, Munich, Germany, and colleagues aimed to investigate whether intraoperative wound irrigation with polyhexanide is effective in reducing surgical site infection within 30 days after open gastrointestinal surgery compared to saline or no irrigation.
For this purpose, they performed IOWISI, a multicenter, 3-armed, randomized clinical trial in 12 university and general hospitals in Germany from 2017 to 2021 with 30-day follow-up. Patients and outcome assessors were blinded to the intervention.
Adult patients undergoing laparotomy were eligible for inclusion. The main exclusion criteria were the inability to provide consent and a clean laparoscopic procedure. Of 11 700 screened, 689 were included and 557 completed the trial; 689 were included in the intention-to-treat and safety analysis.
Patients were allocated in a 3:3:1 ratio to polyhexanide 0.04%, saline, or no irrigation (control) of the operative wound before closure.
Among the 689 patients included, 402 were male and 287 were female. The median age was 65.9 years. Participants were randomized to either wound irrigation with polyhexanide (n = 292), saline (n = 295), or no irrigation (n = 102).
The study’s primary endpoint was surgical site infection within 30 postoperative days, according to the definition of the US Centers for Disease Control and Prevention.
The study led to the following findings:
“Based on the present results, intraoperative wound irrigation with polyhexanide cannot be recommended as standard clinical practice in open clean-contaminated (level of contamination II) visceral surgical procedures,” the researchers wrote.
“More clinical trials are warranted to assess the potential benefit of polyhexanide intraoperative wound irrigation in colorectal, contaminated (level of contamination III) and septic (level of contamination IV) procedures,” they concluded.
Reference:
Mueller TC, Kehl V, Dimpel R, et al. Intraoperative Wound Irrigation for the Prevention of Surgical Site Infection After Laparotomy: A Randomized Clinical Trial by CHIR-Net. JAMA Surg. Published online February 21, 2024. doi:10.1001/jamasurg.2023.7985
Powered by WPeMatico
